Studies have found that clinical research is substantially carbon intensive and more needs to be done to reduce the carbon footprint of clinical trials. One multisite international study, that recruited 10,000 participants, was found to have the same carbon footprint as approximately one person taking 525 round trip flights from London to New York.
The NIHR recently published the latest Carbon Guidelines aimed at researchers and states ‘Research is a core part of the NHS and the National Institute for Health Research (NIHR) must play a role in reducing the carbon emissions from health research.’
The NHS is committed to becoming carbon neutral as soon possible and launched the Greener NHS campaign at the beginning of 2020. Sir Simon Stevens, NHS Chief Executive stated “….we are mobilising our 1.3 million staff to take action for a greener NHS, and it’s why we’ll be working with the world’s leading experts to help set a practical, evidence-based and ambitious route map and date for the NHS to reach net zero.’
Nurses make up the majority of the NIHR research delivery workforce and are key leaders within the organisation. The RCN released a position statement in October 2019 saying ‘Nurses have a duty to protect and promote public health in the face of these threats and have a unique and vital role to play. Their expertise, diverse roles and the trust invested in them mean they can be leaders in protecting the health of the public from the consequences of climate change’. So what could research nurses do to help? Here are 5 suggestions for individual, local and system level actions research nurses could take.
- Take individual action. The global nursing workforce is the biggest healthcare workforce in the world. Nurses taking individual and community action can have a big impact. The Alliance of Nurses for Healthy Environments have begun a new movement called Nurses Drawdown and asks nurses to pledge to taking action in 5 key areas. For more information and to join the movement go to https://envirn.org
- Join a network: such as the Green Nurse Network. This informal network gives nurses an opportunity to connect with other likeminded nurses working to tackle the climate emergency https://networks.sustainablehealthcare.org.uk/network/green-nurse-network Follow them on Twitter @Greennurse.
- Make changes within your scope of professional practice. Changes could include certain elements of practice or whole care pathways. For example, asking study teams to use electronic site files which cut down on paper resource use, cost and also saves storage space. Switching to remote patient study visits, if appropriate, to cut down on patient travel, space and to save patient/staff time. Exploring if study teams can conduct monitoring visits remotely and asking could Site Initiation Visits and study training be done using online methods such as MS teams. These changes could reduce travel emissions, staff time and the associated financial costs.
- Take system level action. You could ask your trust or organisation to publicly declare a climate emergency, if they haven’t done so already. To find out how to do this visit https://healthdeclares.org who can give you information. You could also ask your trust to sign up to the award winning Green Ward Competition, which is a leadership and engagement programme aimed at cutting carbon, improving patient care and saving money, for more information go to https://sustainablehealthcare.org.uk/green-ward-competition
- Education. Learn more about sustainable healthcare. Educate your colleagues about the impacts of climate change on public health. The Nurses Climate Challenge website https://nursesclimatechallenge.org gives information about the role of the nurse and climate change. Learn how to implement environmental changes in practice. The Centre for Sustainable Healthcare offers various practical online courses aimed at healthcare professionals and staff. For more information visit their website https://sustainablehealthcare.org.uk/courses
If you’re interested in discussing sustainable healthcare, join the @SusHealthcare #GreenWard tweetchat on Wednesday 30th September 1pm – 2pm UK time.
Bsc Hons RGN,
Sustainability in Quality Improvement Lead at Centre for Sustainable Healthcare, Oxford, UK.
Former Senior clinical research nurse,
NIHR ALP graduate 2018-19,
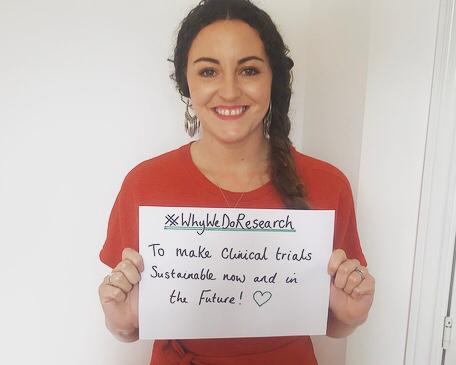






































 RSS Feed
RSS Feed
